Clinical Trial Investigative Site Network Market Projected to Reach USD 18.7 Billion by 2036, With 6.7% CAGR: TMR
Research site consolidation and rapid DCT adoption are reshaping the clinical trial site network market.
Clinical Trial Investigative Site Network Market Growing at 6.7% CAGR Through 2036”
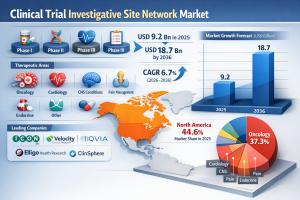
WILMINGTON, DE, UNITED STATES, February 16, 2026 /EINPresswire.com/ -- The global Clinical Trial Investigative Site Network Market was valued at USD 9.2 billion in 2025 and is projected to reach USD 18.7 billion by 2036, expanding at a CAGR of 6.7% from 2026 to 2036. Market growth is driven by the increasing complexity of clinical trials, rising demand for patient recruitment efficiency, and the growing adoption of decentralized and multi-site trial models by pharmaceutical and biotechnology companies.— Transparency Market Research Inc.
The market for the clinical trial investigative site network is projected to expand due to an increase in global clinical research activities, resulting in a greater number of drug and biologics pipelines moving into later-phase studies. The market growth is backed by increasing attention toward efficient patient recruiting and retention, the switch to decentralized and hybrid trial models, as well asthe requirement for standardized, high-quality data collection across various geographies.
Dive Deeper into Data: Get Your In-Depth Sample Now! https://www.transparencymarketresearch.com/sample/sample.php?flag=S&rep_id=86932
The market gains additional support from the growing partnerships amongst sponsors, CROs, and site networks, which work together to enhance trial operations and speed up completion times. The development of patient-focused clinical trial designs creates a need for networks that can effectively connect with and engage their target communities
The clinical trial investigative site network market is undergoing major changes as personalized medicine gains greater acceptance. The specialized operational and clinical requirements for gene therapy, immunotherapy, and other targeted therapies require expertise to support their delivery.
Key Findings of the Market Report
• Oncology remains the dominant therapeutic segment, accounting for approximately 35.1% of market revenue due to the high volume of cancer research globally.
• Phase III trials held the largest market share in 2025, as these large-scale studies involve the highest number of participants and require extensive site coordination.
• Based on end-user, the Contract Research Organization (CRO) segment is witnessing the fastest growth as CROs increasingly partner with site networks to offer "turnkey" clinical trial solutions.
• North America dominated the market in 2025, while Asia-Pacific is projected to be the fastest-growing region through 2033 due to a vast patient pool and lower operational costs.
Global Market: Growth Drivers
• Complexity of Modern Protocols: Transitioning from "one-size-fits-all" drugs to biomarker-driven therapies requires sites with specialized diagnostic capabilities, favoring established networks over standalone clinics.
• Adoption of Hybrid and Decentralized Trials: The integration of telemedicine and wearable tech allows site networks to reach a broader, more diverse patient demographic, significantly improving recruitment timelines.
• Demand for Operational Efficiency: Site networks reduce the administrative burden on sponsors by providing centralized budgeting, contracting, and IRB (Institutional Review Board) submissions.
• Rising R&D Investments: Increased funding for biotechnology startups and a growing pipeline of biologics and biosimilars are necessitating a rapid expansion of qualified investigative sites.
• Focus on Real-World Evidence (RWE): Investigative networks are increasingly used to collect high-quality data in Phase IV (post-marketing) studies to satisfy regulatory demands for long-term safety and efficacy.
Regional Landscape
• North America continues to lead the global market, driven by a mature regulatory environment and the presence of major pharmaceutical giants. In the United States, the FDA’s focus on "Modernizing Clinical Trials" is accelerating the shift toward technology-enabled site networks.
• Asia-Pacific is emerging as a critical hub for clinical research. Countries like China and India are seeing a surge in investigative sites due to favorable government policies and the need to test therapies on a massive, treatment-naive population. This region is becoming a preferred destination for multi-center global trials.
Global Market: Key Players
Major players are expanding their geographic footprints and digital capabilities through aggressive mergers and acquisitions:
• ICON plc
• Velocity Clinical Research
• IQVIA
• Elligo Health Research
• ClinSphere
• ClinChoice
• Access Clinical Trials Inc.
• FOMAT Medical Research INC
• SGS Société Générale de Surveillance SA
• KV Clinical Research
• SMO PHARMINA
• XYLEM RESEARCH LLP
• The Aurum Institute
• Other Prominent Players
Key Developments in Clinical Trial Investigative Site Network Market
• In December 2025, ClinTrial Research entered into partnership with Tarsadia Investments, closing a growth equity funding round aimed at building a next generation clinical site network combining advanced technology and therapeutic leadership.
• In December 2025, Bristol Myers Squibb expanded strategic collaboration with Sarah Cannon Research Institute, thereby enhancing access to clinical trials across the U.S. and strengthening clinical research site partnerships.
• In July 2025, Rakuten Medical expands patient recruitment and site network, actively recruiting at more than 15 clinical sites in the U.S., Taiwan, and Japan, with planned expansion to Eastern Europe for supporting its global Phase III oncology trial.
• In January 2025, Advarra launched its Study Collaboration solution, a platform designed to accelerate study startup by automating workflows, thereby enhancing real time visibility and improving collaboration amongst sponsors, CROs, and site teams. This solution integrates study start-up and enrollment planning tools to streamline site activation.
Elevate Your Business Strategy! Purchase the Report for Market-Driven Insights: https://www.transparencymarketresearch.com/checkout.php?rep_id=86932<ype=S
Market Segmentation
By Therapeutic Area
• Oncology
• Cardiology
• Central Nervous System (CNS)
• Pain Management
• Endocrine & Metabolic Disorders
• Others
By Phase
• Phase I
• Phase II
• Phase III
• Phase IV
By End-User
• Pharmaceutical & Biopharmaceutical Companies
• Contract Research Organizations (CROs)
• Medical Device Companies
By Region
• North America
• Europe
• Asia-Pacific
• Latin America
• Middle East & Africa
More Trending Reports by Transparency Market Research –
Endovascular Treatment Devices Market - https://www.transparencymarketresearch.com/endovascular-treatment-devices-market.html
Home Diagnostics Market - https://www.transparencymarketresearch.com/home-diagnostics-market.html
DNA Methylation Market - https://www.transparencymarketresearch.com/dna-methylation-market.html
Enteral Feeding Devices Market - https://www.transparencymarketresearch.com/enteral-feeding-devices-market.html
Hypodermic Needles Market - https://www.transparencymarketresearch.com/hypodermic-needles-market.html
About Transparency Market Research
Transparency Market Research, a global market research company registered at Wilmington, Delaware, United States, provides custom research and consulting services. Our exclusive blend of quantitative forecasting and trends analysis provides forward-looking insights for thousands of decision makers. Our experienced team of Analysts, Researchers, and Consultants use proprietary data sources and various tools & techniques to gather and analyses information.
Our data repository is continuously updated and revised by a team of research experts, so that it always reflects the latest trends and information. With a broad research and analysis capability, Transparency Market Research employs rigorous primary and secondary research techniques in developing distinctive data sets and research material for business reports.
Contact:
Transparency Market Research Inc.
CORPORATE HEADQUARTER DOWNTOWN,
1000 N. West Street,
Suite 1200, Wilmington, Delaware 19801 USA
Tel: +1-518-618-1030
USA – Canada Toll Free: 866-552-3453
Website: https://www.transparencymarketresearch.com
Email: sales@transparencymarketresearch.com
Abhishek Budholiya - Marketing Team
Transparency Market Research Inc.
+1 518-618-1030
email us here
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.